|
12/28/2023 0 Comments Hollow cathode lamp block diagram 
The sample vapour is atomized by the vapour impacting on a glass bead. Some form of venturi where the air sucks in the sample liquid. The mechanism for sample introduction includes: For magnesium, a magnesium hollow cathode lamp is used. 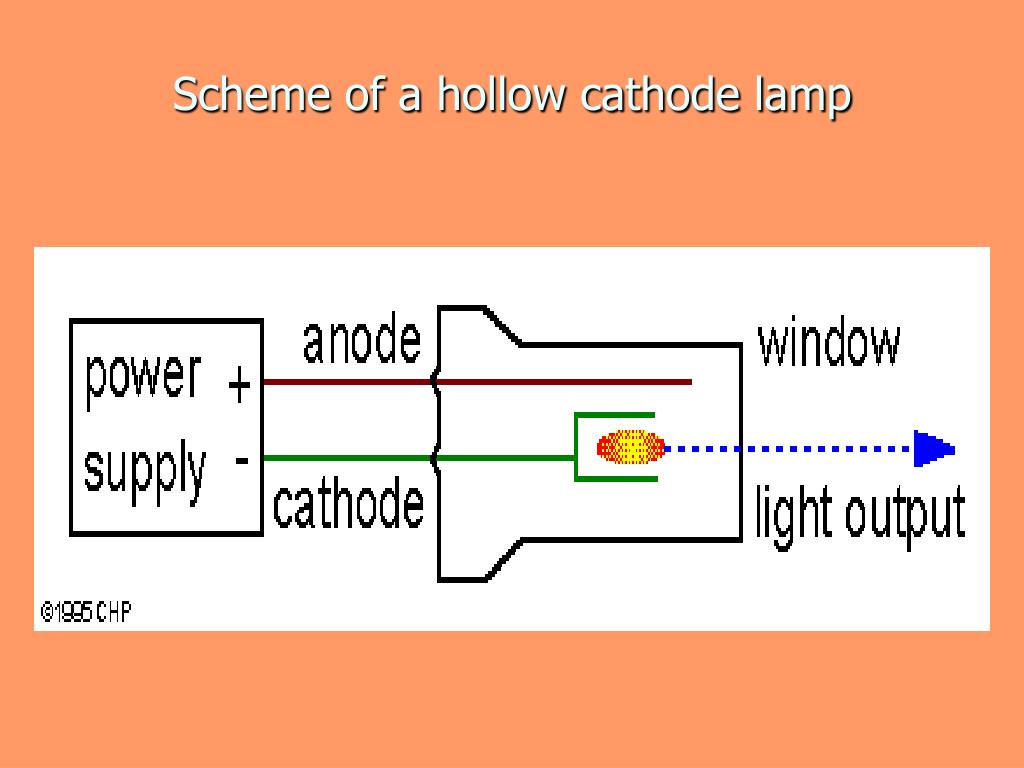
That is, if the determination is for Copper, then a copper hollow cathode lamp was used. The specific cathode is made of that metal. To analyse for different metals, requires a different hollow cathode lamp for each metal of interest. The wavelength sources evolved to become Hollow Cathode Lamps, where the metal of interest is incorporated into the cylindrical cathode and the anode and cathode are sealed in a glass envelope.Ī voltage is applied between the electrodes and the fill gas ions are attracted to the metal cathode and their impact releases the metal wavelengths required for the absorption in the flame, of that specific metal. The signal strength after the flame then decreases with increasing concentration. The greater the concentration of metal atoms in solution, then the more photons that are absorbed in the flame, from the wavelength source. This photon does not fall on the amplifier sensor and the signal strength is reduced by the photon loss. The electron ABSORBS this wavelength photon and lifts to the next energy level. The wavelength source provides the wavelength energy, exactly, that is required by the ‘excited’ electron. 
The theory of operation is that the aspirated metal atoms electrons are ‘excited’ by the flame energy but do not have the necessary energy to ‘jump’ to the next quantum energy level. This allowed the sensor to measure the wavelength intensity before and after the flame. The sensor was synchronized with a chopper set up between the wavelength source and the flame. The sensor measured the signal before the flame was aspirated with the unknown metal in solution and then again after the sample was aspirated into the flame. A photon amplifier sensor on the other side of the flame, then measured the signal through the flame. Walsh’s solution was to irradiate the low temperature flame with the metal wavelength of interest. The Bunsen burner temperatures were too cool for significant emission line strengths. His dilemma was that, trying to observe the metal emission lines from metal electron quantum transitions, was only observable for the Group 1, alkali elements such as Sodium, 589.0nm. He continued his research to find a fast alternative spectroscopic analysis to the slow colorimetric method for metal identification. He developed a number of methods for rapid spectrographic analysis of metals.Īfter the war ended, he accepted a position at CSIRO’s, Fishermen’s Bend laboratories in Melbourne. His work included determination of metal composition from the debris of enemy bombers. After a period of time, the atoms move to the glass walls of the lamp or back to the surface of the cathode.In 1940 Alan Walsh was a young physicist working at the British Non Ferrous Metals Laboratories during World War II. The release of this radiation allows for data to be collected and information gathered pertaining to the metal of interest. Some of these metal atoms in the cloud have moved to an excited energy state and as they return to their ground state, they emit a specific radiation which is characteristic to the metal of interest. The process of producing this cloud is called sputtering. The removal of these atoms produces a cloud around the cathode. If the voltage applied is large enough, the ions of the inert gas gather enough energy to remove some of the metal atoms of interest from the surface of the cathode by striking the surface. This current allows for electrons to move to the anode and cathode. This voltage is applied across the anode and the cathode and generates a current of 5-15 mA. Ionization is the process by which neutral atoms are converted to charged species. First, the inert gas contained inside of the glass tube is ionized by a voltage. The excitation process of the element of interest takes place through a few steps. Hollow-cathode lamps in an atomic absorption spectrometer 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
